VenaSeal® - Closure: Results Over 6 Years Treatment. A follow-Up Study Conducted on 1950 Truncal Saphenous Veins in 1061 Cases
Ulf Th.Zierau
DOI10.21767/2573-4482.18.03.17
Ulf Th.Zierau*
Founder and CEO of SAPHENION®- Surgeon, Vascular Surgeon, Phlebologist, Endovascular Specialist Artery, Endogenous Specialist Berlin/Rostock, Germany
- *Corresponding Author:
- Ulf Th.Zierau
Founder and CEO of SAPHENION®-Surgeon
Vascular Surgeon, Phlebologist
Endovascular Specialist Artery
Endovenous Specialist Berlin/Rostock
Germany
Email: dr.zierau@yahoo.de
Received Date: July 06, 2018; Accepted Date: July 11, 2018; Published Date: July 27, 2018
Citation: Zierau UT (2018) VenaSeal® - Closure: Results Over 6 Years Treatment. A follow-Up Study Conducted on 1950 Truncal Saphenous Veins in 1061 Cases. J Vasc Endovasc Therapy. 3:17.
Abstract
We report about 6-years results of a retrospective comparative study of VenaSeal®- Closure in the treatment of 1950 saphenous veins (1349 GSV, 517 SSV, VSAL in 53 cases, VSAM in 30 and Giacomini vein in 2 cases). Treatment included also ulcera crures in 11 cases.
The present research paper sheds light on the advantages and disadvantages and presents the 72-month results of a single-center ambulatory clinical study with retrospective design.
Keywords
Saphenous veins; Truncal varicose veins; Sonography; Therapeutic; Anesthesia
Introduction
In the base, all varicose veins should be treated actively. This we can find in nearly all guidelines worldwide. All the specialists know that mobilisation and compression alone cannot normalize the venous function of outflow venous blood from the leg. An insufficient varicose vein is working like a downpipe-the blood pressure at the lower leg is increased chronically. And so we get the typical chronique venose disease. Nearly 70% of all adults in europe have clinical signs of this CVD.
Since 18 years by now, varicosis has been increasingly treated endovenously. Before this, the varicose veins were treated radically with the "stripping"-method, a 110 years old radical surgery method.
At the start, the rather inconvenient VNUS® Closure Plus procedure and the more convenient linear laser procedure were used, and these were followed in 2006/2007 by the bipolar RFITT® catheter, the VNUS® Closure Fast system and the radial laser. Thus, in the course of the last few years, plenty of experience has been gathered with endoluminal therapy, quality criteria have been defined and standards for the different techniques have been developed [1-7].
One very important technical development combined with the beginning of the endovenous therapy was the colour ultrasound (duplex)-we can see the catheter inside the veins, the glue and we can control the tip of catheter, the work inside the vessel and the effects inside the body-without any radiation and without intra venous contrast agents. These is a very important fact, because working with an endovenous catheter without ultrasound isn`t a fully noninvasive therapy because of using phlebography. Ratzek et al. have described exactly the sonographic appearances of common disorders of all tissues. They have worked about the high sensitivity of ultrasound in tissue diagnostics [8-10].
In addition, 15 years ago, far from the beaten tracks of radio wave and laser, the development of a fascinatingly simple, yet nevertheless highly effective method of sealing veins-the VenaSeal® closure technique-was initiated. After CE approval had been granted in the autumn of 2011, a number of vein centers in Germany and Europe started using the VenaSeal®-system.
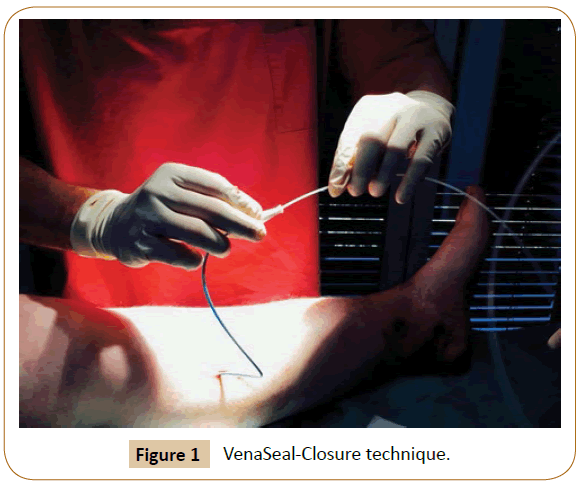
By now, 40 centers are working successfully with the new therapy system in Germany alone. Today there is an approval in all countries, also in USA since 2/2015. The author has applied Venaseal for the first time in a great saphenous vein on 1st August 2012 (Figure 1).
Materials and Method
Based on the manufacturer's application instructions, sealing with the VenaSeal-system was started 1-3 cm from the saphenofemoral junction, and a spot of glue was applied at intervals of 2-3 cm, depending on the diameter and the flow/pressure of the vein. Thick branch-offs of auxiliary side branches were additionally treated with single-shot glue. The maximal diameter of treated truncal veins was 2-3 cm also venous aneurysmas were treated. The follow-up observation period in our study was up to 72 months.
The great saphenous vein was treated in 1349 cases, in 517 cases the small saphenous vein was treated and in 76 cases the trunc of an inguinal accessoric vein was sealed. Two Giacomini veins also were treated.
VenaSeal® interventions were performed under light sedation with Dormicum or local anesthesia for venous access accompanied by music therapy, 91 patients didn’t got any anaesthesia. One patient performed pain acupuncture on herself on point G4.
All patients are given a follow-up examination by duplex sonography in the scope of a prospective study (our own quality management) on the 1st/14-30th/70-90th day as well as after 6 and 12 months. After this we controlled every following year. All duplex sonography examinations post interventions were done by another colleague, not by the vascular surgeon treated the truncal veins (Figure 2).
Results
During the time period from 1st August 2012 to 31st July 2018 (72 month), Venaseal® was applied to achieve closure of the vein in 1950 truncal varicose veins. In 324 patients one saphenous vein were treated; in 606 patients two saphenous veins were treated in 109 patients 3 saphenous veins were treated. In 20 cases 4 truncal veins and in each 1 case 5 and 6 truncal veins were treated simultaneously.
Grade 2-3 saphenous varicosis of the GSV according to Hach, and in the case of the SSV Grade 2 saphenous varicosis according to Hach, was the inclusion criterium. In accessoric veins we treatet the inguinal trunc in length between 12-25 cm.
On the 1st day 1950 veins were checked (1938 veins were closed initially=99.4 %) in the scope of follow-up, and up to the 30th day, partial recanalization was found in 37 veins, and complete recanalization was found in 9 veins. This corresponds to a closure rate of 97.74% Over a time period of 3 months up to 4 month after the treatment, we were able to follow up 1544 saphenous veins (79.3% of all veins that had been treated), and here we found 39 partial and 13 complete recanalizations. The closure rate is thus 97.34%.
1332 saphenous veins (68.2%) were followed up over a 6-month time period, and 45 partial and 25 complete recanalization’s were found, resulting in an effectiveness of 96.41%.
No further recanalization’s were found after 6 years. In the followup period of 4 years after therapy we controlled 1204 truncal varicose veins (61.7%) up to now. All 11 leg ulceras were healed until to 11 weeks after intervention.
1950 truncal varicose veins having been sealed with Venaseal®, the results achieved over the entire time period of 72 months are equivalent to a closure rate of 96.41%. The pain score for subjectively felt pain on the 1st. day post-surgery (range 1-10) was between 1.6 and 3.4 (2.1)-in RFITT 3.8 and 4.1). In 156 treated veins (8%), we observed a postoperative unspecific inflammatory skin reaction after approx. 10-14 days in the Venaseal group; with appropriate antiphlogistic treatment with ibuprofen and cooling dressings, this subsided within 3-5 days.
In all other cases subjected to follow-up examinations, no complications of any kind, no paresthesias or hypesthesias, no permanent skin reactions, no phlebitis or thrombosis or infections were observed. Only in 5 cases we were seen a lymphatic fistula at the peripherial punktion.
In particular, even subcutaneously situated saphenous veins could be glued without any significant skin reaction (reddening, swelling).
We also clearly prefer VenaSeal® in treatment of SSV and now also in GSV due to the large number of neurological sensations in connection with treatment by Laser and Radiofrequency [11,12].
Nearly all patients were greatly surprised at the fully ambulatory intraoperative procedure and the brief and pleasant postoperative convalescence phase (Figure 3).
All patients were able to leave the office between 30 and 120 minutes after the intervention.
In the case of VenaSeal, we have up to now refrained from applying compression therapy in over 90% of all cases. We prefer to use compression stockings only in cases, the diameter of the treated vein it’s over 1.5 cm or in treatment of venous aneurysm or ectatic varicose veins.
Discussion
In the last 18 years, the necessary quality criteria’s for endovascular interventions on veins with varicose changes were largely laid down, and several comparative studies on functional efficiency of radical stripping surgery on the one hand and endovenous treatments on the other hand were furthermore conducted. By now, it has emerged as an undeniable fact that endovenous interventions do not only exhibit a merely cosmetic advantage as was hitherto assumed. They also have clinical advantages and quite significantly reduce side effects and complications such as still occur regularly today as in the past in connection with the conventional surgical technique.
Thus, the colleagues who work with endovenous procedures meanwhile have reliable criteria for a high-quality therapy [1,6,12-15].
The VenaSeal® closure procedure is the newest technical development in the series of endovenous therapeutic procedures. Although it is a catheter - based procedure in terms of the basic principle of the therapeutic approach, it differs fundamentally with regard to the closure technique. While the glue likewise gives rise to a certain temperature (approx. 45°C-50°C), the procedure is not a thermal one. Side effects as those known to occur in connection with laser and radio wave therapy ultimately play no significant role here. The necessary reliable closure is achieved by means of a cyanoacrylate superglue, the basic chemical formula of which has been known since several decades, and which is being used in neuroradiology in the treatment of vascular malformations since 1981. We also worked with this glue since 1988 in vascular surgery at the Charitè-hospital.
We do not need anesthesia anymore and can in most cases do without postoperative compression therapy. Elastic stockings should nevertheless by all means be recommended after the treatment of thicker saphenous varicose veins measuring > 1.2 cm, and they become compulsory where we intend to apply gluing therapy in larger lumens measuring 1.5 cm and more.
The significantly reduced side effects and a well-nigh negligible pain score are also clear advantages in comparison with laser and radio wave therapy. No paresthesias, no hypesthesias, no phlebitis, extremely rare occurrence of skin pigmentations are only a few of the important advantages of the VenaSeal®- procedure. In the final analysis, the new procedure has to meet solely the hard criterion of efficacy, namely the permanence of an effective vein closure. And as far as this aspect is concerned, both the first results of the eSCOPE study (14) and the results of single-center studies, and also currently of the VeClose study [14] are very good. The closure rate is similarly high as that achieved with radio waves, namely between 93%-100% when all results are summarized [15].
Thus, the VenaSeal®-procedure appears to be on the same level with, or even superior to the high - frequency radio wave system [5,16]. In the time periods between 6 and 12 months covered by follow - up examinations up to now, both procedures have proven quite clearly superior (99.6%) [13,15,16] to laser therapy in terms of effectiveness.
The results of first comparative studies show that the VenaSeal®- glue is clearly superior with regard to postoperative side effects though. Both the pain score and the rate of side effects are very low in comparison [12]. Particularly pain as well as the neurological side effects no longer play any significant role at all.
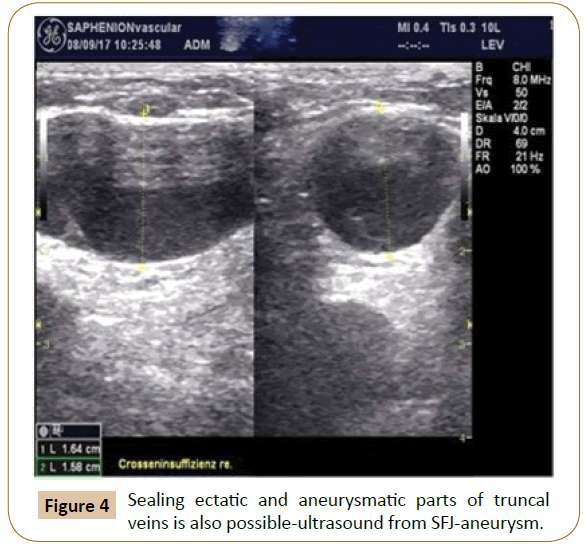
These are the main problem associated with laser and radio wave therapy though. By now, VenaSeal® has undeniably become at SAPHENION the therapy of first choice for the treatment of the SSV. Here, we meanwhile consider the well - known risk of neurological side effects and complications associated with application of the laser and radio frequency techniques as being too high [3-6,13-16]. In the light of the 17 years of experience we have gathered by now, we recommend that every vein center that applies endovenous treatment should have at least 2 alternative treatment procedures at its disposal. For us, this means that in practical work with VenaSeal®, all insufficient saphenous veins should as far as possible always be treated in one session (Figure 4).
Independently of this, we at SAPHENION® meanwhile regard Venaseal®-closure as treatment of first choice in the range of catheter-supported therapeutic procedures for GSV and SSV or VSAA-varicosis [17-23].
Conflict of Interests
There are no conflicts of interest; the present research paper was not sponsored.
References
- Creton D, Rea B, Pittaluga P, Chastanet S, Allaert FA (2012) Evaluation of the pain in varicose vein surgery under tumescent local anaesthesia using sodium bicarbonate as excipient without any intravenous sedation. Phlebology 27: 368-373.
- Elias S, Raines JK (2012) Mechanochemical tumescentless endovenous ablation: Final results of the initial clinical trial. Phlebology 27: 67-72.
- Huisman LC, Bruins RMG, van den Berg M, Hissink RJ (2009) Endovenous laser ablation of the small saphenous vein: respective analysis of 150 patients, a cohort study. Eur J Vasc Endovasc Surg 38: 199-202.
- Almeida JI, Javier JJ, Mackay EG, Bautista C, Proebstle TM (2015) One-year follow up of first human use of cyanoacrylate adhesive for treatment of saphenous vein incompetence. J Vasc Surg Venous Lymphat Disord 3: 125.
- Proebstle TM, Vago B, Alm J, Göckeritz O, Lebard C, et al. (2008) Treatment of the incompetent great saphenous vein by endovenous radiofrequency powered segmental thermal ablation: First clinical experience. J Vasc Surg 47: 151-156.
- Proebstle TM, Alm J, Rasmussen L, Dimitri S, Whiteley M , et al. (2012) Cyanoacrylate adhesive for treatment of great saphenous vein incompetence without tumescent anesthesia and without compression therapy abstract presented to the american college of phlebology annual meeting 2012 hollywood, Florida.
- Rosen RJ, Contractor S (2004) The use of cyanoacrylate adhesives in the management of congenital vascular malformations. Sem Interv Radiol 21: 59-66.
- Abdel Razek AA, Al Belasy F, Ahmed W, Haggag M (2015) Assessment of articular disc displacement of temporomandibular joint with ultrasound. J Ultrasound 18: 159-163.
- Razek AA, Fouda NS, Elmetwaley N, Elbogdady E (2009) Sonography of the knee joint. J Ultrasound 12: 53-60.
- Razek AA, El-Basyouni SR (2016) Ultrasound of knee osteoarthritis: Interobserver agreement and correlation with Western Ontario and McMaster Universities Osteoarthritis. Clin Rheumatol 35: 997-1001.
- Zierau U Th (2014) VenaSeal® - Therapie bei SAPHENION - 18 Monats - Ergebnisse an 379 Stammvenen; Vortrag auf dem.
- Zierau U.Th (2016) Venenverklebung versus radiofrequenztherapie bei varicosis-verlaufsstudie über 36 monate mit 1139 behandlungen. Vasomed 28: 212-216.
- Gibson K, Morrison N, Kolluri R, Vasquez M, Weiss R, et al. (2018) Twenty-four month results from a randomized trial of cyanoacrylate closure versus radiofrequency ablation for the treatment of incompetent great saphenous veins. J Vasc Surg Venous Lymphat Disord 6: 606-613.
- Morrison N and Gibson K (2015) Veclose Study: Preliminary Month 1 Data.
- Proebstle TM, Alm J, Rasmussen L, Dimitri S, Lawson JA , et al. (2013) The european multicenter study on cyanoacrylate embolization of refluxing great saphenous veins without tumescent anaesthesia and without compression therapy. J Vasc Surg Venous Lymphat Disord 1: 101.
- Rasmussen LH, Bjoern L, Lawaetz M, Lawaetz B, Blemings A, et al. (2010) Randomised clinical trial comparing endovenous laser ablation with stripping of the great Saphenous vein: Clinical outcome and recurrence after 2years. Eur J Vasc Endovasc Surg 39: 630-635.
- Almeida JI, Min RJ, Raabe R, McLean DJ, Madsen M (2011) Cyanoacrylate adhesive for the closure of truncal veins: 60-day swine model results. Vasc Endovasc Surg 45: 631-635.
- Almeida JI, Mackay EG, Bautista C, Proebstle T (2012) Cyanoacrylate glue great saphenous vein ablation: preliminary 180-day follow up of a first-in-man feasibility study of a no- compression-no-local anaesthesia technique. J Vasc Surg 55: 297.
- Keel D, Goldman MP (1999) tumescent anesthesia in ambulatory phlebectomy: Addition of epinephrine. Dermatol Surg 25: 371-372.
- Lawson J, Gauw S, Vlijmen CV, Pronk P, Gaastra M, et al. (2013) Sapheon: The solution? Phlebology 28: 2-9.
- Min RJ, Almeida JI, McLean DJ, Madsen M, Raabe R (2012) Novel vein closure procedure using a proprietary cyanoacrylate adhesive: 30-day swine model results. Phlebology 27: 398-403.
- Shepherd AC, Gohel MS, Lim CS, Hamish M, Davies AH (2010) The treatment of varicose veins: an investigation of patient preferences and expectations. Phlebology 25: 54-65.
- Shadid N, Ceulen R, Nelemans P, Dirksen C, Veraart J, et al. (2012) Randomized clinical trial of ultrasound-guided foam sclerotherapy versus surgery for the incompetent great saphenous vein. Br J Surg 99: 1062-1070.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences