Shall We Think in Using Endoanchors in all EVAR and TEVAR Procedures?
Paulo Eduardo Ocke Reis and Andrés Reyes Valdivia
DOI10.21767/2573-4482.20.05.4
1Department of Specialized and General Surgery, Fluminense Federal University, Rio de Janeiro, Brazil
2Department of Vascular and Endovascular Surgery, Ramón y Cajal’s University Hospital, Madrid, Spain
- *Corresponding Author:
- Paulo Eduardo Ocke Reis
Department of Specialized and General Surgery
Fluminense Federal University, Rio de Janeiro, Brazil
Tel: +55 21 2629-5000
E-mail: Paulo Eduardo Ocke Reis
Received Date: April 27, 2020; Accepted Date: May 01, 2020; Published Date: May 08, 2020
Citation: iOcke Reis PE, Valdivia AR (2020) Shall We Think in Using Endoanchors in all EVAR and TEVAR Procedures? J Vasc Endovasc Therapy Vol.5 No.2:4
Editorial
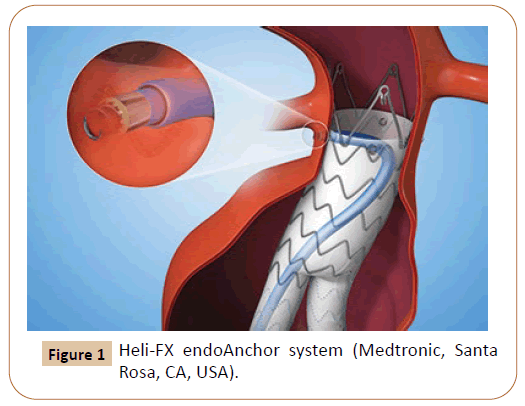
Parodi first described in 1991 the exclusion of an abdominal aortic aneurysm (AAA) from circulation with endovascular abdominal aortic repair (EVAR) and since then, many studies have shown that when the best seal zone between the endograft and the aortic wall was achieved, the best outcomes were encountered [1]. The approach to treat complex aortic thoracic or abdominal aneurysmatic disease remains a surgical challenge and different techniques have been used, current projects are focusing on outcome and device evaluation with EVAR or thoracic endovascular aortic repair ( TEVAR ) to avoid endoleaks, especially type I endoleaks. More recently, the Heli‐FX EndoAnchor System (Medtronic, Santa Rosa, CA, USA) (Figure 1) has been used to prevent endoleak in aneurysms with challenging proximal or distal neck anatomies improving fixation where they are deployed. [2-4].
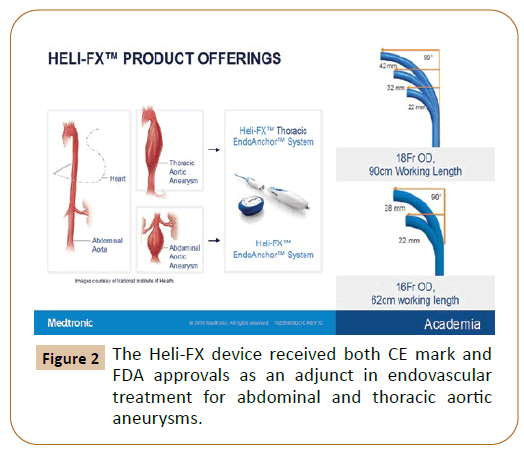
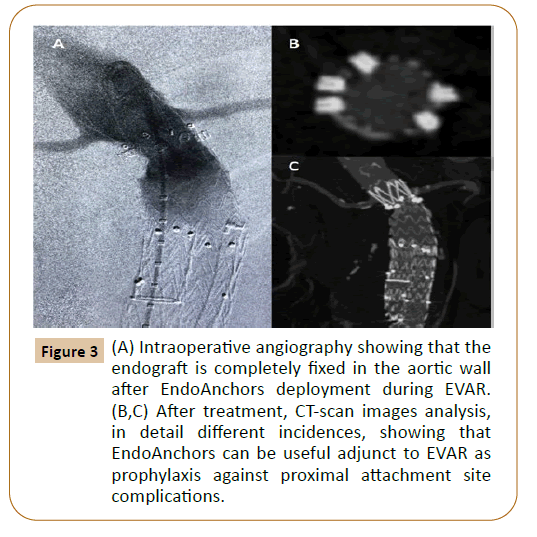
The Heli-FX system success strictly depends on adequate positioning of the endograft and then, achieving a perpendicular aortic wall penetration. Other criteria to succeed on the therapy, as in every endovascular procedure, relies on the learning curve experience of the work team [4]. Reyes Valdivia et al. found a higher risk failure in arch deployments, probably attributable to the small learning curve experience in this location; however, a high rate of adequate aortic wall penetration was achieved in the descending thoracic aorta, where EndoAnchors proved useful for distal endograft fixation [4]. One study derived from the ANCHOR registry provides evidence of no penetration of the EndoAnchors into the aortic wall, being this condition the only predictive factor of post procedural type IA endoleak [5]. Thus, we might ask ourselves if a more liberal use of EndoAnchors for EVAR and TEVAR procedures seems logical at this time. The Heli-FX device received both CE mark and FDA approvals as an adjunct in endovascular treatment for abdominal and thoracic aortic aneurysms (Figure 2). After treating 109 patients with 674 EndoAnchors at two centers in different aortic anatomies, Reyes Valdivia et al concluded the use of EndoAnchors to be safe and effective in complex aortic aneurysms anatomies (Figure 3) [6]. However, a better understanding of the device is still needed. The ANCHOR registry, a sponsored study, has already published on some reports the benefits, regarding sac regression and delayed aortic neck dilatation, when adequately used. Despite these encouraging results, more real-world data is required to better understand the bad and the good of the device. We need more research and further studies that support their use. If these are achieved in the near future, we definitely see a clear win for the use of EndoAnchors in those anatomies where proved to be beneficial.
Figure 3: (A) Intraoperative angiography showing that the endograft is completely fixed in the aortic wall after EndoAnchors deployment during EVAR. (B,C) After treatment, CT-scan images analysis, in detail different incidences, showing that EndoAnchors can be useful adjunct to EVAR as prophylaxis against proximal attachment site complications.
References
- Parodi JC, Palmaz J, Barone H (1991) Transfemoral intraluminal graft implantation for abdominal aortic aneurysm. Ann Vasc Surg 5: 491-499.
- Kasprzak P, Pfister K, Janotta M, Kopp R (2013) EndoAnchor placement in thoracic and thoracoabdominal stent-grafts to repair complications of nonalignment. J Endovasc Ther 20: 471-480.
- Ongstad SB, Miller DF, Panneton JM (2016) The use of EndoAnchors to rescue complicated TEVAR procedures. J Cardiovasc Surg 57: 716-729.
- Reyes Valdivia A, Busto Suárez S, Duque Santos Á, Zanabili Al-Sibbai AA, Gandarias Zúñiga C, et al. (2020) Evaluation of Endoanchor aortic wall penetration after thoracic endovascular aortic repair. J Endovasc Ther 27: 240-247.
- Goudeketting SR, van Noort K, Ouriel K, Jordan Jr WD, Panneton JM, et al. (2018) Influence of aortic neck characteristics on successful aortic wall penetration of EndoAnchors in therapeutic use during endovascular aneurysm repair. J Vasc Surg 68: 1007-1016.
- Valdivia AR, Santos AD, Guaita JO, Zúñiga CG, Chaudhuri A (2019) Can Endoanchors be Used in Any Endovascular Aortic Procedure? Lessons Learned After 674 Endoanchors in 109 Patients in a Two-centre Experience. Eur Vasc Endovascular Surg 58: e227.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences